This clinical trial is sponsored by Novartis Pharmaceuticals Corp.
This clinical trial is sponsored by Novartis Pharmaceuticals Corp.
The cycle of chronic hives, also called chronic urticaria, can seem like it occupies every part of life. When the itching keeps coming back, it can feel like a never-ending struggle, even with treatment. That is why we are conducting a trial for those diagnosed with chronic spontaneous urticaria (CSU).
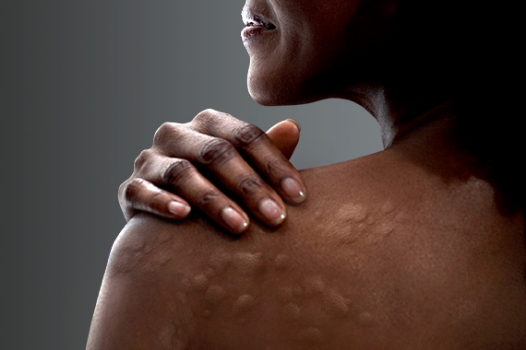
This clinical trial will evaluate the efficacy of remibrutinib in comparison to dupilumab in treating adult participants who have chronic spontaneous urticaria (CSU) that is not adequately controlled with antihistamines. The study drug is an oral medication taken twice a day and is being compared to an injection given every two weeks.




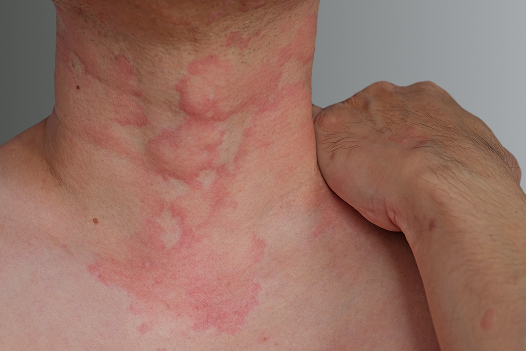
Chronic Spontaneous Urticaria (CSU), also known as chronic hives, is characterized by itchy welts on the skin daily or nearly every day for at least six weeks. Chronic hives can be very uncomfortable and interfere with sleep and daily activities.¹
To see if you may qualify for this trial, take the online questionnaire.






If you are interested in joining, the first step is to take a brief questionnaire and see if you meet the initial qualifications. You won’t need to submit your contact information to get your results. If you qualify, you may be contacted by a trial team representative for further evaluation.
A trial team representative will contact you ONLY after you have agreed to be contacted.
Once you have passed the online pre-screener and granted permission to be contacted, a trial team representative will call you. During this initial phone call, they may ask additional screening questions and confirm the answers you submitted online. If you qualify, the next step is to set up an in-person visit to a local site and begin collecting your medical records.
At your appointment, the study doctor and research team will explain all the details about the study, including possible risks and what participation involves. They will also answer any questions you may have.
The treatment period for this study will last 12 weeks. During this time, you will receive either remibrutinib, taken twice per day orally, or dupilumab given as an injection every two weeks. Neither you nor the study doctor will know which medication group you belong to. You will also take a stable daily dose of an antihistamine, which will be provided by the study doctor.
During the study, you will need to visit the trial site up to eight times. You will also be asked to answer questions about your symptoms in a daily diary. You may be reimbursed for reasonable expenses that you may incur as a result of taking part in this study. These expenses may include parking, meals, transportation or other travel-related expenses.
Clinical studies involve volunteers and help researchers learn how to better diagnose, treat, and prevent various diseases or conditions. These studies might explore new medicines, combinations of medications, surgical procedures, devices, or innovative ways to utilize existing treatments4.
The goal of this study is to evaluate the efficacy of remibrutinib in comparison to dupilumab for the treatment of chronic spontaneous urticaria (CSU).
Novartis is the trial sponsor.
Throughout the trial, all participants will have their health monitored in several health checks, which include physical examinations, heart activity checks (ECG) and blood and urine tests with the trial team.
Reimbursement may be offered to cover expenses incurred from your participation in the study. Please speak with the study coordinator at the site regarding the possible reimbursements offered.
You do not have to pay for the treatment or trial procedures that are not part of your routine medical care. However, you may be responsible to pay for routine medical care that may be performed during the trial, some of which may be covered by your insurance.
You are free to stop participating in this study at any time and for any reason.
The first step to trial participation is to take the online questionnaire. You will be notified immediately if your answers match the key study requirements. If you qualify online, a member of the trial team will follow up with additional information about your qualification.
1. Maurer, Marcus, et al. “Antihistamine‐Resistant Chronic Spontaneous Urticaria Remains Undertreated: 2‐Year Data from the AWARE Study.” Clinical & Experimental Allergy, vol. 50, no. 10, Sept. 2020, pp. 1166–75, "https://onlinelibrary.wiley.com/doi/10.1111/cea.13716"
2. Hsieh, Jane, and Jason K Lee. “Chronic Spontaneous Urticaria.” CMAJ : Canadian Medical Association Journal = Journal De L’Association Medicale Canadienne, U.S. National Library of Medicine, 16 Jan. 2017, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5235931/.
3. “Chronic Spontaneous Urticaria: What to Know.” WebMD, WebMD, https://www.webmd.com/skin-problems-and-treatments/features/urticaria-chronic-spontaneous.
4. Clinical Trials - About Clinical Studies. Mayo Clinic. Published 2026. https://www.mayo.edu/research/clinical-trials/about-clinical-studies
We do not sell data to third parties. Data you submit is handled according to our Privacy Policy and web site use agreements.
To control data you have already submitted, or to log a do not sell data request, make a data request here.
To control data you have already submitted, make a data request here.
Data you submit is handled according to our Privacy Policy and web site user agreements.